Genentech
Optimal Patient Pathway Study
Mapping what it’s like to live with Idiopathic Pulmonary Fibrosis.
Overview
Genentech needed to understand what patients with Idiopathic Pulmonary Fibrosis — a rare, progressive, and ultimately terminal lung disease — experience across their entire care journey. Not clinical outcomes. Lived experiences.
Alongside one other researcher, I designed and conducted in-depth, semi-structured interviews in patients' homes across four US regions. We followed participants from the first symptom they dismissed as allergies or a cold, through years of misdiagnosis, into disease management and end-of-life planning.
Role
Research lead
Methods & artifacts
Contextual inquiry
Empathy maps
Coded transcripts
Patient personas
Journey maps
“I’ve just felt like I’ve been flying by the seat of my pants. Stuff happens and you think — okay, is that because of the disease? Is that because of the medication?”
— Jackie, age 74, IPF patient
In the field
Why we went to people’s homes
A clinical interview captures what a patient reports. A home visit captures what they live. We chose the latter deliberately. Being in someone's home — seeing the furniture rearranged to accommodate an oxygen equipment, the medications lined up on the kitchen counter, the family member quietly listening from the doorway — gave us context that no survey or clinic-based interview could.
We recruited participants from four distinct regions: urban and rural, coastal and inland. The geographic spread was intentional. Access to ILD Centers of Excellence varies enormously by zip code, and we needed to understand whether proximity to specialized care changed how patients understood and navigated their diagnosis.
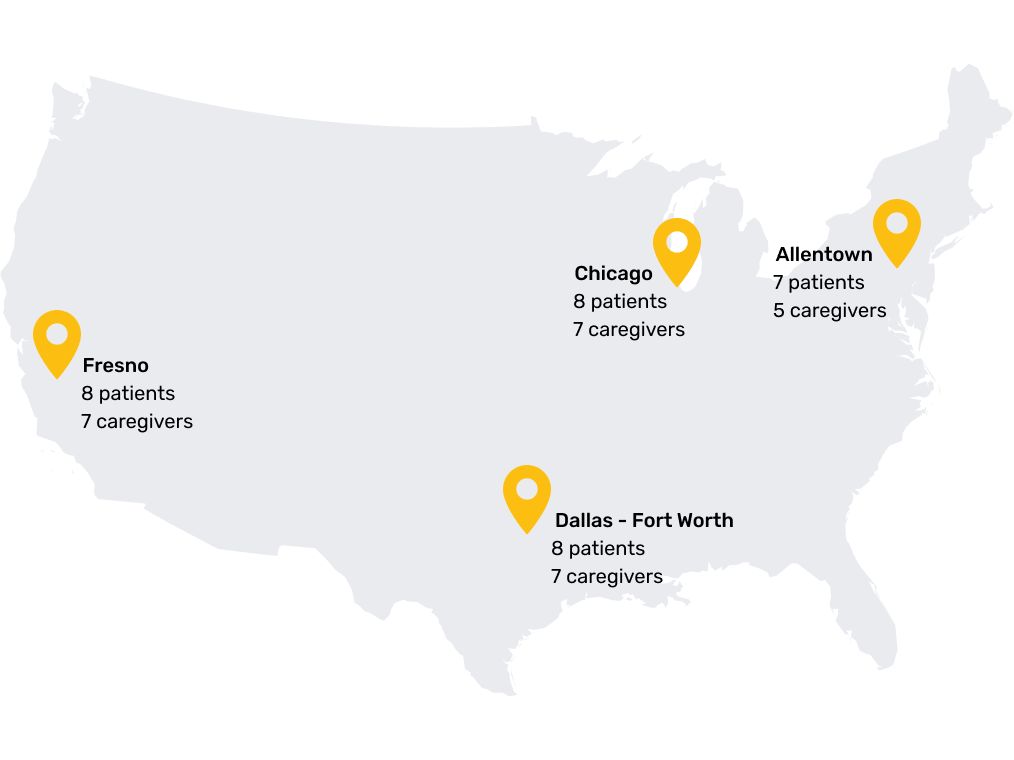
Study participants were recruited from four regions spanning urban and rural communities — deliberately mapped to capture variation in access to ILD Centers of Excellence.
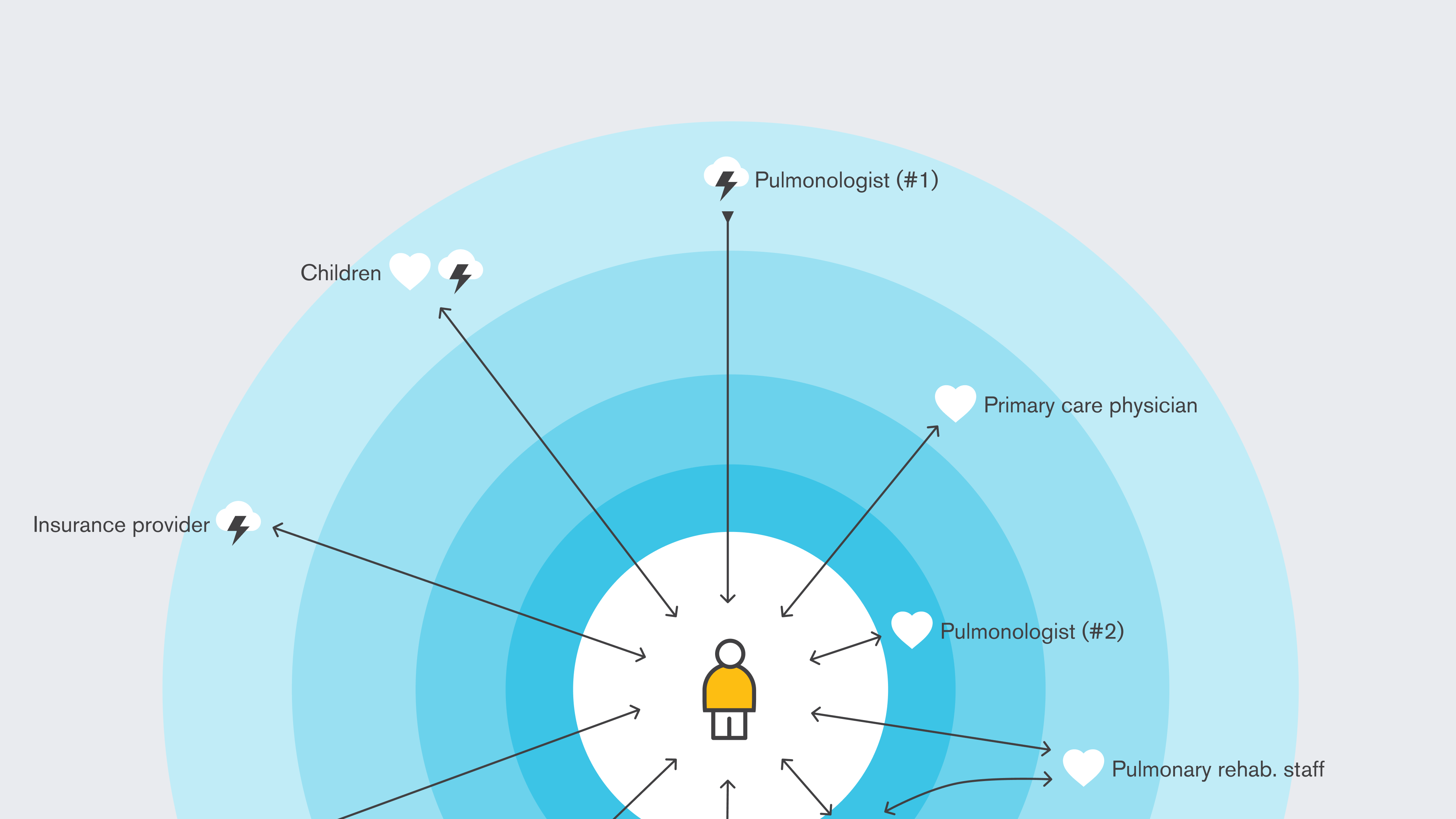
At each session’s end, I led a circles-of-care exercise where patients mapped those involved in their care and ranked them by closeness and trust. Spouses occupied the center, specialists and nurses near the center, and on the periphery, initial primary care physicians and specialists – who were often viewed as lacking empathy.
Patients mapped their care relationships by closeness. Across all participants, initial primary care providers and specialists consistently sat at the periphery.
Synthesis
From 70+ hours of conversation to patterns
Two researchers. 31 interviews. Tens of thousands of words of transcript. The synthesis work took longer than the fieldwork.
Using a systematic coding process, tagging meaningful statements, tracking frequency, and mapping relationships between codes, we uncovered something deeper than a list of complaints. What emerged was a portrait of systemic uncertainty: patients who struggled to describe their symptoms to doctors, who didn’t know what questions to ask, and who felt as though they were improvising their way through a terminal diagnosis.
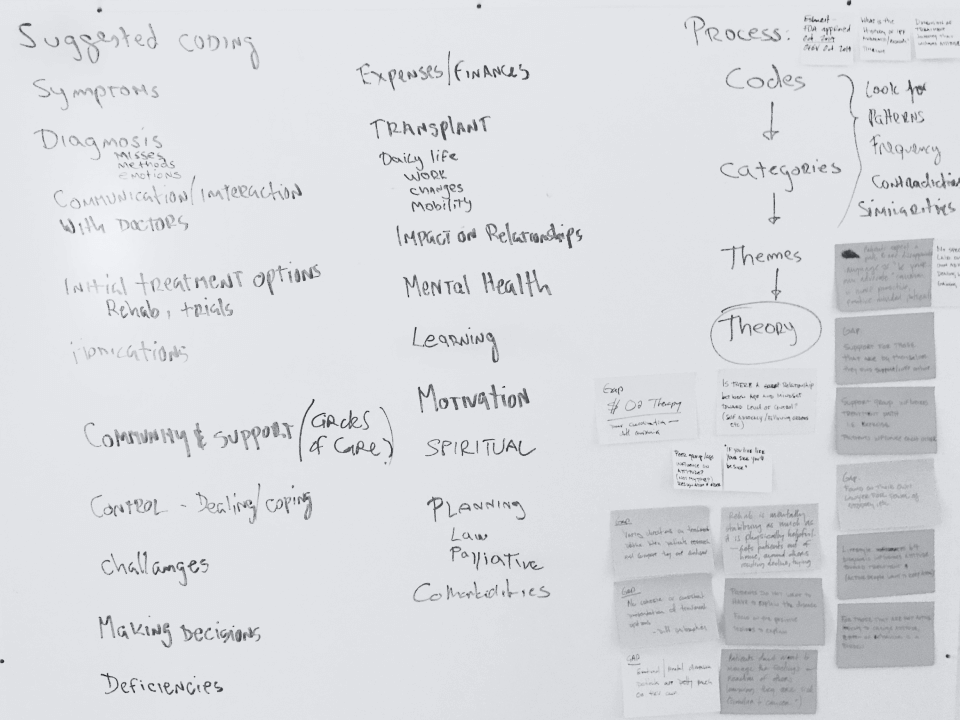
Codes clustered by theme during synthesis sessions.

Every transcript tagged with emergent themes.
From the coded data, we developed four patient personas and one caregiver persona — each grounded in a specific combination of demographics, disease stage, geography, and psychographic profile. I then built comprehensive journey maps for each, tracing the pathway from the first symptom dismissed as aging through to transplant evaluation or end-of-life care planning.

Persona documents and journey maps for each patient type — capturing not just demographics and situational factors, but the emotional register at every stage of the IPF experience.
Our research found urgent gaps in IPF care
01
Diagnosis is profoundly delayed
On average, patients saw 3–5 physicians over 2+ years before receiving an accurate IPF diagnosis. Misdiagnoses weren't outliers, they were the norm.
02
Patients are more willing than assumed
Contrary to clinical assumptions, patients were open to treatment. What they lacked was clear information.
03
Multidisciplinary teams were a bright spot
Patients who encountered team care during lung transplant evaluation reported their most positive healthcare experiences. This model wasn't consistently available.
04
Community and peer support matter enormously
IPF support groups were cited across nearly every interview as a primary source of information and emotional support. Often more valuable to patients than formal care visits.
Opportunities where the system can do better
_
Earlier specialist access
Introducing an IPF team — including social workers — at working diagnosis rather than confirmed diagnosis could reduce years of uncertainty.
_
Grassroots physician education
IPF is rare enough that primary care physicians rarely recognize it. Targeted education of PCPs, radiologists, and community health workers offers the highest leverage for earlier detection.
_
Standardized care pathways
ILD Centers of Excellence vary widely in protocol. Training and guideline support from the Pulmonary Fibrosis Foundation could raise the floor, especially in rural regions.
_
Telemedicine for access gaps
Rural patients were most isolated from specialist care. Telemedicine — for both patients and the radiologists who first see suspicious imaging — could meaningfully shorten the path to diagnosis.
Reflection
This was the most emotionally demanding research I‘ve done. You can‘t sit in someone's living room while they talk about end-of-life planning and remain purely analytical. The challenge was carrying that emotional weight through a long synthesis process without letting it distort the findings — and using it, instead, as motivation to make the deliverables actually useful to the people making decisions about IPF care. The personas and journey maps were presented to Genentech's clinical team to inform future program development.